Background:
Classic myeloproliferative neoplasms (MPN) polycythemia vera (PV), essential thrombocythemia (ET), and myelofibrosis (MF), are characterized by concurrent risk of thrombotic and hemorrhagic complications. These risks are reported to be further compounded perioperatively ( Blood, 2008), though scarce contemporary data exist. Further, the lack of evidence-based guidelines leads to heterogeneous management. The objective of this study was to comparatively assess perioperative complication rates (thrombotic, hemorrhagic, and survival) and management practices in a large MPN population.
Methods:
Consecutive surgical interventions in patients with WHO-defined PV, ET and MF recruited from the Quebec MPN Research Group Registry (6 centers; between 1986-2023) were analyzed. Endpoints (90 days post-surgery) and surgical definitions were per convention. Standard statistics were used (JMP® Pro 14.1.0 software; SAS Institute, Cary, NC, USA).
Results:
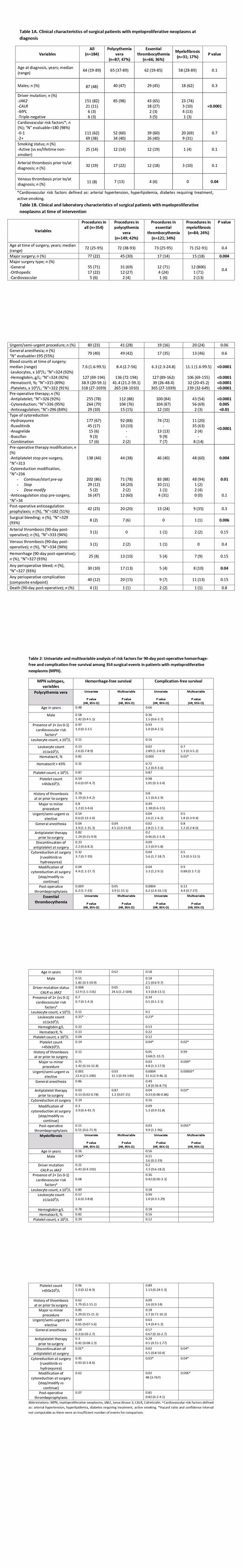
A total of 354 procedures were captured in 184 patients: PV, n=87 (47%); ET, n=66 (36%); MF, n=31 (17%). Cardiovascular risks were balanced; previous venous thrombosis was enriched in PV (p=0.04; Table 1A). Major surgeries were performed in 45 cases (30%), primarily PV (p=0.004); most being general (69%) (Table 1B). Interventions were urgent in 41 cases (28%); 49 (42%) of 195 evaluable were under general anesthesia. The majority of PV/ET subjects continued antiplatelet and cytoreductive agents peri-intervention, though cytoreduction was stopped in 12-22%. Thromboprophylaxis was administered in n=42 (23%). Surgical bleeding occurred in n=8 (2%). At 90-day follow-up, arterial/venous thrombosis occurred each in n=3 (1%), hemorrhage in n=25 (8%), and death in n=4 (1%). Overall complication rate was 12% (n=40). PV patients displayed significantly higher rates of surgical bleeding (6%; p=0.0006) and perioperative hemorrhage (13%; p=0.04).
Factors impacting 90-day surgical outcomes:Hemorrhage-free survival (HFS): PV: General anaesthesia (p=0.04) and antithrombotic prophylaxis (p=0.05) negatively impacted HFS on multivariate testing (Table 2). ET: Driver mutation status CALR vs JAK2 (HR 24.6; 95% CI 1.2-504; p=0.05) and urgent interventions (HR 11.5; 95% CI 0.93-144; p=0.03) emerged as independent predictors for HFS. MF: Discontinuation of antiplatelet agents perioperatively was the sole factor impacting HFS (p=0.01). Perioperative complication-free survival (CFS; composite endpoint): PV: While several factors were significant on univariate testing (procedure, leukocytosis, ruxolitinib exposure, cytoreduction modification, thromboprophylaxis), multivariate testing disclosed only lower hematocrit (< 35%; p=0.05) to impact complication rates. ET: Urgent (p<0.0001) and major procedures (p=0.009), thromboprophylaxis use (p=0.001), platelet count > 450 x 10 9/L (p=0.02), and absence of antiplatelet (p=0.02) were significant independent predictors of CFS. MF: Antiplatelet discontinuation (HR 6.5; 95% CI 0.8-50.4; p=0.02), nature of cytoreduction (ruxolitinib vs hydrea; p=0.04) and cytoreduction discontinuation pre-operatively (p=0.006) negatively impacted CFS on multivariate analysis. Limited informative events for thrombosis/survival precluded analyses.
Conclusions:
This is, to our knowledge, the largest and most contemporary analysis of surgical procedures in patients with MPN. Firstly, it confirms prevalent bleeding perioperatively, though balanced by fewer thrombotic events, calling for further study of thromboprophylaxis/antiplatelet practices in this setting. Second, it discloses key management patterns, notably routine discontinuation of cytoreduction, which had a detrimental impact on outcomes. Third, correlation of uncontrolled platelets and overzealous hematocrit control with higher complication rates in ET and PV, respectively, underscores importance of judicious target control. Further, CALR driver mutation status associated with bleeding risk in ET, independent of platelet count, suggesting additional contributing mechanisms. Finally, ruxolitinib use was an independent predictor of perioperative complications in MF, though this may reflect skewing towards higher-risk populations. Further studies will be required to validate findings, with this data serving as a preliminary signal for awareness of risk and reappraisal of management strategies.
Disclosures
Busque:BMS: Honoraria; Novartis: Honoraria; Taiho: Honoraria; Paladin: Honoraria. Sirhan:GSK: Honoraria; Novartis: Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal